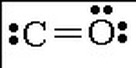Carbon Monoxide
Chapter 6 Carbon Monoxide compound CO
01. Summary:
In chapter 6 Carbon Dioxide was the subject. Due to the chemical in
the highly requested automobiles of that time period. It killed and
injured many New Yorkers. Anyone who had the unlucky chance to inhale
the drug was killed or became deathly sick.
02. State of matter at room temperature (20o C) Solid/liquid/gas-Properties with reference to COMPRESSABILITY, VOLUME, SHAPE, KINETIC ENERGY
State of matter: Gas
03. Chemical/Physical properties-categorize as intensive/extensive
Intensive-
Extensive-
04. Classify matter-pure substance vs. mixtures
05. Bond type with explanation (If covalent, is it polar or nonpolar and explain)
06. Lewis structures to illustrate bonding
01. Summary:
In chapter 6 Carbon Dioxide was the subject. Due to the chemical in
the highly requested automobiles of that time period. It killed and
injured many New Yorkers. Anyone who had the unlucky chance to inhale
the drug was killed or became deathly sick.
02. State of matter at room temperature (20o C) Solid/liquid/gas-Properties with reference to COMPRESSABILITY, VOLUME, SHAPE, KINETIC ENERGY
State of matter: Gas
- High compressibility
- Low density
- High kinetic energy
- Volume depends on container
03. Chemical/Physical properties-categorize as intensive/extensive
Intensive-
- Density:1.15 kg/m^3
- Color: colorless
- Boiling point: -312.7 F (-191.5 C)
Extensive-
- Molar mass: 28.01 g/mol
04. Classify matter-pure substance vs. mixtures
- Pure substance
- Compound
05. Bond type with explanation (If covalent, is it polar or nonpolar and explain)
- Covalent
- Polar because different atoms have different electronegative
06. Lewis structures to illustrate bonding