Radium
Chapter 8 Radium element
01. Summary:
In Chapter 8 radium a familiar name in the book Norris was brought
back into play. He was brought in to help with yet another health
hazard Radium. Instead of civilians scientist were the victims. In
France and other countries radium was an accidental killer. To keep it
under wraps politicians swayed the medical examiners but in the end
the medical examiners and politicians got in some major trouble, and
now medical examiners and politicians don't do under the table
business anymore.
02. State of matter at room temperature (20o C) Solid/liquid/gas-Properties with reference to COMPRESSABILITY, VOLUME, SHAPE, KINETIC ENERGY
State of mater : solid
03. Family (description of characteristics)/Group-
Group 2
Family: The alkaline earth metals
reactive metals
distinctive flame colours
two electrons in their outermost s subshell. general harder, denser, and have higher melting points than their alkali metal.
04. Chemical/Physical properties-categorize as intensive/extensive
Intensive
Extensive
05. Classify matter-pure substance vs. mixtures
06. Electron configuration-
[Rn]7s2
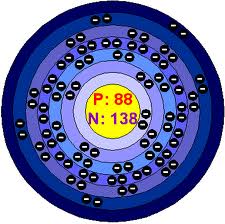
07. Bohr model-
01. Summary:
In Chapter 8 radium a familiar name in the book Norris was brought
back into play. He was brought in to help with yet another health
hazard Radium. Instead of civilians scientist were the victims. In
France and other countries radium was an accidental killer. To keep it
under wraps politicians swayed the medical examiners but in the end
the medical examiners and politicians got in some major trouble, and
now medical examiners and politicians don't do under the table
business anymore.
02. State of matter at room temperature (20o C) Solid/liquid/gas-Properties with reference to COMPRESSABILITY, VOLUME, SHAPE, KINETIC ENERGY
State of mater : solid
- Low compressibility
- High density
- Low kinetic energy
- Volume constant
- Shape depends on container
03. Family (description of characteristics)/Group-
Group 2
Family: The alkaline earth metals
reactive metals
distinctive flame colours
two electrons in their outermost s subshell. general harder, denser, and have higher melting points than their alkali metal.
04. Chemical/Physical properties-categorize as intensive/extensive
Intensive
- Boiling point: 2,084°F (1,140°C)
Extensive
- Atomic mass:226
05. Classify matter-pure substance vs. mixtures
- Pure substance
- Element
06. Electron configuration-
[Rn]7s2
07. Bohr model-
08. Lewis Dot-