Wood Alcohol
Chapter 2 Wood Alcohol compound CH3OH
01. Summary:
In chapter 2 of the poisoners handbook Wood Alcohol was a silent
killer. In the streets of New York it claimed the lives of many
civilians. Doctors were accustomed to alcohol poisoning by then, the
routine of life in the Prohibition era. The bootlegged whiskies and
so-called gins often made people sick. The liquor produced in hidden
stills frequently came tainted with metals and other impurities. But
this outbreak was bizarrely different. The deaths, as investigators
would shortly realize, came courtesy of the U.S. government and
murderers.
02. State of matter at room temperature (20o C) Solid/liquid/gas-Properties with reference to COMPRESSABILITY, VOLUME, SHAPE, KINETIC ENERGY
State of matter- liquid
03. Chemical/Physical properties-categorize as intensive/extensive
Intensive-
Extensive-
04. Classify matter-pure substance vs. mixtures
05. Bond type with explanation (If covalent, is it polar or nonpolar and explain)
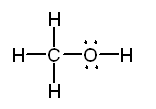
06. Lewis structures to illustrate bonding
01. Summary:
In chapter 2 of the poisoners handbook Wood Alcohol was a silent
killer. In the streets of New York it claimed the lives of many
civilians. Doctors were accustomed to alcohol poisoning by then, the
routine of life in the Prohibition era. The bootlegged whiskies and
so-called gins often made people sick. The liquor produced in hidden
stills frequently came tainted with metals and other impurities. But
this outbreak was bizarrely different. The deaths, as investigators
would shortly realize, came courtesy of the U.S. government and
murderers.
02. State of matter at room temperature (20o C) Solid/liquid/gas-Properties with reference to COMPRESSABILITY, VOLUME, SHAPE, KINETIC ENERGY
State of matter- liquid
- Low compressibility
- Medium kinetic energy
- Volume constant
- Shape depends on container
03. Chemical/Physical properties-categorize as intensive/extensive
Intensive-
- Color: colorless
- Odor: alcohol like
- Boiling point:148.1 F (65 C)
- Density: 791.80 kg/m^3
- Melting point: -144.4 F (-98 C)
Extensive-
- Molar mass: 32.04 g/mol
04. Classify matter-pure substance vs. mixtures
- Pure substance
- Compound
05. Bond type with explanation (If covalent, is it polar or nonpolar and explain)
- Covalent
- Polar because different atoms have different electronegative
06. Lewis structures to illustrate bonding