Methyl Alcohol
Chapter 7 Methyl Alcohol compound CH4O
01. Summary:
In Chapter 7 Methyl Alcohol was the topic. It started out as a topic
only for mayors and officials to debate about, but soon turned to a
topic that whispered through the streets of New York. On Sunday
Morning, March 20, 1927, Ruth Snyder found her mother dead after a
home invasion in which she was knocked unconscious. After her story
didn't add up she blamed her lover Judd Gray. Through evidence and
some careless mistakes the couple was found guilty and sentenced to
the electric chair.
02. State of matter at room temperature (20o C) Solid/liquid/gas-Properties with reference to COMPRESSABILITY, VOLUME, SHAPE, KINETIC ENERGY
State of matter: liquid
03. Chemical/Physical properties-categorize as intensive/extensive
Intensive
Extensive
04. Classify matter-pure substance vs. mixtures
05. Bond type with explanation (If covalent, is it polar or nonpolar and explain)
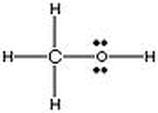
06. Lewis structures to illustrate bonding
01. Summary:
In Chapter 7 Methyl Alcohol was the topic. It started out as a topic
only for mayors and officials to debate about, but soon turned to a
topic that whispered through the streets of New York. On Sunday
Morning, March 20, 1927, Ruth Snyder found her mother dead after a
home invasion in which she was knocked unconscious. After her story
didn't add up she blamed her lover Judd Gray. Through evidence and
some careless mistakes the couple was found guilty and sentenced to
the electric chair.
02. State of matter at room temperature (20o C) Solid/liquid/gas-Properties with reference to COMPRESSABILITY, VOLUME, SHAPE, KINETIC ENERGY
State of matter: liquid
- Low compressibility
- Medium kinetic energy
- Volume constant
- Shape depends on container
03. Chemical/Physical properties-categorize as intensive/extensive
Intensive
- Boiling point:149 F (65 C)
- Melting point:-144.4 F (-98 C)
- Density :791.80 kg/m^3
Extensive
- Molar mass:32.04 g/mol
04. Classify matter-pure substance vs. mixtures
- Pure substance
- Compound
05. Bond type with explanation (If covalent, is it polar or nonpolar and explain)
- Covalent
- Polar because different atoms have different electronegative
06. Lewis structures to illustrate bonding