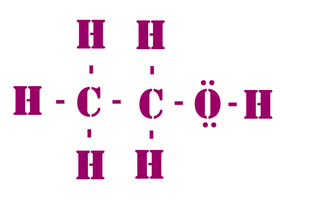Ethyl Alcohol
Chapter 9 Ethyl Alcohol compound C2H5OH
01. Summary:
The chapter starts off with records from Alexander O. Gettler. Case 1
a moderate drinker, with good health just up and died from alcohol
poisoning. In case 2 a woman who drank a half of pint of whiskey was
found dead lying on her back. Both deaths got chemists thinking, they
wondered how people of good health and and a ripe age could die so
suddenly. Then they git it, they had been killed by alcohol because
people usually forgot the risk of it. By the 1930s Gettler has
assembled an encyclopedia list of cases for his research into the
chemistry of drunkenness. He's researched in labs and even tested his
experiments and findings on animals. By the end of his experiments he
along with other scientist discovered that alcohol poisoning was
dangerous and could be considered as a poison because of some of
ingredients.
02. State of matter at room temperature (20o C) Solid/liquid/gas-Properties with reference to COMPRESSABILITY, VOLUME, SHAPE, KINETIC ENERGY
State of matter: liquid
03. Chemical/Physical properties-categorize as intensive/extensive
Intensive
Extensive
04. Classify matter-pure substance vs. mixtures
05. Bond type with explanation (If covalent, is it polar or nonpolar and explain)
06. Lewis structures to illustrate bonding
01. Summary:
The chapter starts off with records from Alexander O. Gettler. Case 1
a moderate drinker, with good health just up and died from alcohol
poisoning. In case 2 a woman who drank a half of pint of whiskey was
found dead lying on her back. Both deaths got chemists thinking, they
wondered how people of good health and and a ripe age could die so
suddenly. Then they git it, they had been killed by alcohol because
people usually forgot the risk of it. By the 1930s Gettler has
assembled an encyclopedia list of cases for his research into the
chemistry of drunkenness. He's researched in labs and even tested his
experiments and findings on animals. By the end of his experiments he
along with other scientist discovered that alcohol poisoning was
dangerous and could be considered as a poison because of some of
ingredients.
02. State of matter at room temperature (20o C) Solid/liquid/gas-Properties with reference to COMPRESSABILITY, VOLUME, SHAPE, KINETIC ENERGY
State of matter: liquid
- Low compressibility
- Medium kinetic energy
- Volume constant
- Shape depends on container
03. Chemical/Physical properties-categorize as intensive/extensive
Intensive
- Odor: alcohol like
- Color: colorless
- Boiling point: 78.5°C (173.3°F)
- Density : 789.00 kg/m³
Extensive
- Molar mass: 46.06844 g/mol
04. Classify matter-pure substance vs. mixtures
- Pure substance
- Compound
05. Bond type with explanation (If covalent, is it polar or nonpolar and explain)
- Covalent
- Polar because different atoms have different electronegative
06. Lewis structures to illustrate bonding